新常科学的人生观穹顶之下
New normal scientific perspective “under the dome”
文/壮歌德 翻/董阳
Throughout history people spend their lives dedicated to consumption. They consume to survive and to enjoy, planning consumption. They compare their consumption with that of other people, their neighbors, and various role models.
A human can be likened to an amoeba which seeks out good things and then engulfs them. Conventional economic wisdom promotes more “consumption” as a panacea that will benefit us all. From ancient philosophy and modern science, this perspective is hideously incorrect.
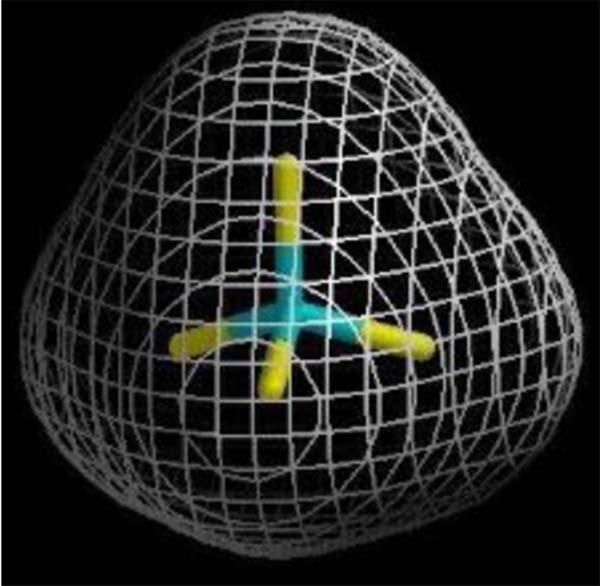
At this scale the carbon atom is scarce, 0.6 in solids including all biomass, and 0.9 in the air. Yet the hexagonal carbon atom is the key to life trading bonds with hydrogen for bonds with oxygen.
Before the Industrial Revolution had set in to change the planet, and before Dalton discovered atoms (1802), the foundations of economics were in already place, analyzing the cycle between farm produce and cottage industry (Quesnay 1756) and the monetary exchange calculation of consumer items by primitive hunters (Smith 1776). The human perspective of consumers and planners, world leaders was governed by money values of price, income and GDP.
Objective observation and computation using recent advances in science can zoom from personal activities up to global conditions, and also down from eyeball senses to the interaction of atoms. What a person eats can be linked to global food demand and down to the chemistry of carbohydrates.
From a global omniscient perspective, life appears as a quest to manipulate carbon-hydrogen bonds to break and for the carbon to attract oxygen from the air to form new bonds. The bond formed is double the strength of the carbon-hydrogen bond. Chemists have known this for decades and used it to label the “calories” of foods and also then same “energy available” in all fuels including the big 3 fossil fuels, coal, oil and natural gas.
Simply, the carbon-hydrogen bond has a strength of about 4 electronvolts and that of carbon-oxygen is over 8 ev. When a kilogram the simple molecule CH4 (natural gas) reacts with O2 the difference in total bond strengths is 26 megajoules, the key fact that heats rooms, cooks food and drives machines.
C-H bond strength 4 ev C-O bond strength 8 ev
|
In the new perspective using science to understand economic activity, the reason that locking carbon into a stronger bond (carbon dioxide) actually releases energy for use. Modern science in the past 2 decades has been able to reveal the topological nature of atoms in molecules (Bader 1991) and the bond strengths can be set against the context of the overall molecular electron charge density within the molecule’s shape.
This benefit has been obvious (without being understood) as long as life existed and evolved into human consciousness as a love for food and all forms of energy, including draft animals, wood fires and eventually fossil fuels.
Now concern with the global economy, the financial crises, debt so great as to be incomprehensible and at the same time the idea that human activity has impact on climate, it requires a fresh perspective. Economist, financial experts and world leaders transfixed with money measurement and remedies must be introduced to the reality that money no longer represents.
Frustrated with the inability to link money uniformly to reality the respected journal The Economist, introduced “the Big Mac index” 巨无霸指数. To simplify, a bread bun index 馒头指数 of uniform carbohydrate content of 100 kilojoules can be viewed as a store of carbon-hydrogen bonds that are consumed by a worker and transformed into “work”.
To assist economists understand, each carbon-hydrogen bond can be valued as two halves of 50 yuan, and bonded together constitute a value of 50 yuan. In the envelope of the carbohydrate molecule there is also “dangling loose” one half of a 100 yuan note. The same situation is in an oxygen molecule, bonded by halves of 50 yuan notes, but with a spare half of 100 yuan. On collision the bonds break and a new molecule of carbon dioxide is formed with the halves of 100 yuan notes as bonds.
But the envelope of total molecular charge is constant and the “energy not needed” is released. The dictionary defines “entropy” as “unavailable energy” and the molecular envelope in the products of the process, dominated by the tightly locked carbon dioxide bond is high entropy.
An objective alien observer may well wonder why humans seem hellbent on a race to increase high entropy under the dome 穹顶之下. Our past lives have focused on the energy released that drives economic processes, from individual consumption of food up to the huge inputs of fossil fuels for industry. Now that drive can be envisaged in a universal currency of carbon-hydrogen bonds, metaphorically intended to “make” 200 yuan for life and a better life out of breaking bonds worth 50 yuan.
We now must recognize this also as a race to stuff another 200 yuan into choking the air under the dome we live in.
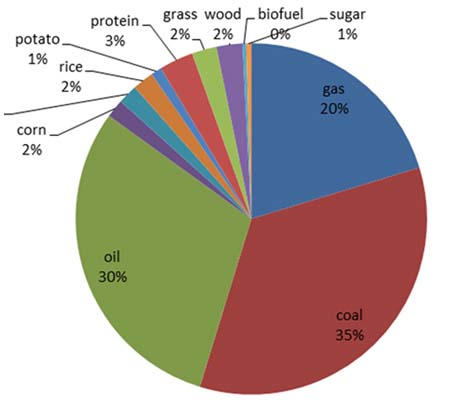
If a mantou costs 50 cents then the standard carbon-hydrogen bond is worth 6.64 x 10-24 fen (RMB). The C-H bonds traded in 2014 totaled 1,799 x 1037, and by this approach total human activity is “worth” (in “mantou reality bonds”) $20 trillion.The nominal global GDP last year according to conventional economic accounting was $80 trillion. The carbon-oxygen bonds produced by human effort was $40 trillion according to the mantou approach.
Number of carbon-hydogen bonds (x 1037) in the hydrocarbons and carbohydrates that were traded for carbon-oxygen bonds in 2014.
新常态科学角度看世界:穹顶之下
New normal scientific perspective “under the dome”
文/壮歌德 John Coulter 译/董阳
近期著名媒体人柴静出品的纪录片《穹顶之下》引发各方关注,激起人们对空气污染这一环境问题的热烈讨论。
消费需求的膨胀,导致生产快速增长,诱发此类环境问题。其实,纵观历史,人类的存在就是一个持续消费的过程。人们靠消费来存活和享受生命,并不断同邻居以及各种各样的名人比较消费水平。
人类如同阿米巴变形虫,遇到好东西就一口吞掉。传统经济学认为“消费”惠及普罗大众,是促进社会进步的灵丹妙药。然而,从古典哲学和现代科学的角度来看,这种观点都是极其错误的。
生物圈中的原子数目估算:
| 不同元素的原子 | 数目(×1040) | ||
| 液态 | 气态 | 固态 | |
| 氧原子 | 4466 | 1003 | 67.7 |
| 氢原子 | 8932 | ||
| 氮原子 | 4530 | ||
| 硅原子 | 22.6 | ||
| 铝原子 | 6.7 | ||
| 铁原子 | 2.0 | ||
| 钠原子 | 2.9 | ||
| 钙原子 | 2.2 | ||
| 锰原子 | 1.9 | ||
| 钾原子 | 1.7 | ||
| 碳原子 | 0.9 | 0.06 | |
人们通过重组物质的方式来消费。理论上,地球可用的物质总量用原子数目来表示的话,总数为19041 x 1040。其中海洋中氢原子个数为8932 x 1040,海洋中的氧原子个数为4466 x 1040,空气中的氧原子个数为1003*1040,岩石和植被中的氧原子个数为68 x 1040。从这一角度来看,碳原子数量非常少,固体中仅含0.06 x 1040个,涵盖所有的生物质,而气体中包含0.9 x 1040个。而且,在人类将碳氢键转化为碳氧键的过程中,起关键作用的只有六角形碳原子。
工业革命的兴起,改变了我们的地球。在那之前,在道尔顿于1802年发现原子之前,经济学的基础就已经成型。当时的经济学家分析农产品和家庭手工业之间的周期性关联(魁奈,1756年),并计算远古时代猎人消费涉及的货币交换问题(史密斯,1776年)。当时,消费者和各国领袖关注的焦点都集中在价格的价格、收入和GDP的货币价值上。
近几年科学不断发展,人们得以采取更为客观的方式进行观察和计算。大到全球状况,小到个人活动,上至肉眼可见范围,下至原子活动,都可以观察到。这些进展可将个人饮食,同全球性食物需求以及碳水化合物的化学反应联系起来。
从地球上空俯瞰,生命的存在似乎是这样一个过程:尝试各种方法断开碳氢键,以使碳原子同空气中的氧原子结合形成碳氧键。新形成的碳氧键键能是碳氢键的两倍。数十年前,化学家就发现了这一现象,并借此计算食物的“卡路里”和各类燃料中的“可用能”,包括三大化石燃料煤炭、石油、天然气在内。
简单来讲,单个碳氢键的键能约为4ev,单个碳氧键的键能在8ev以上。当一千克CH4分子(即天然气)同O2发生化学反应时,反应前后两种化学键之间的键能差总量为26兆焦,这部分能量可用来取暖、做饭和驱动器械。
碳氢键的键能 4ev 碳氧键的键能 8ev
当前,研究者开始应用现代科学来解释经济活动。从这一角度来看,将碳氢键转化为键能更大的碳氧键(二氧化碳)的过程会释放能量。过去二十年里,现代科学的进展,使得科学界观察到分子中原子的拓扑性质(巴德,1991年)。与此同时,键能的含义可扩展为:分子内部电子电荷相互作用的一部分。
一个CH4分子中四个碳氢键的键能总和为17ev,分子总能量为122ev。该分子同氧原子结合后,能量总和为471ev。此结合过程产生二氧化碳和水,而能量总和维持不变。然而,由于二氧化碳中的碳氧键更为稳定,因而多余能量释放出来,为人类所用。此为近年来出现的新型研究方法,其有效性有待进一步探究。
CH4 电子密度图
虽然远古时期的人类并不了解这一过程的原理,但其带来的明显好处激发出人们对食物和各种形式的能源(包括牲口、火和化石燃料)的热爱。
著名杂志《经济学人》发现无法将金钱同现实统一联系起来,故而引进“巨无霸指数”这一概念。考虑到中国国情,此处用馒头指数替代。简要来说,含100千焦能量的均一碳水化合物的“馒头指数”,可被视为一组碳氢键的集合。工人消耗这一集合并将其转化为“工作”。当前,全球经济、金融危机、不可思议的巨额债务、人类活动对气候造成的影响引发广泛关注,人们需要从全新的视角看待这些问题。以往,经济学家、金融专家和各国领袖习惯了用货币衡量一切,如今他们必须面对现实,承认金钱再也不能代表一切。
经济学家可参考下图的化学键转换过程,以理解上述说法。假设每个碳氢键的价值为一张撕成两半的50元纸币,总价值为50元。在碳水化合物分子内悬挂着半张100元,氧分子内化学键也是一张撕成两半的50元纸币,但还包含半张游离的100元纸币。当碳水化合物和氧气发生化学反应时,原有化学键断裂形成新的碳氧键,键能价值100元。
如果一个外星人正在观察地球,他会很疑惑为什么人类在拼命推高生物圈的熵值。过去,小到个人饮食、大至工业对化石燃料的巨额消耗,我们致力于释放能量,以促进经济进步。然而,若将碳氢键看做通用货币,则人们的各项活动都可比作破坏50元来“拼凑”200元的过程。但分子电荷的总和是恒定的,反应过程释放出分子“不需要”的能量。字典中对“熵”的定义即为“不可用的能量”。上述过程生成的主要化合物为二氧化碳,其分子内部的化学键十分牢固,熵值很高。
现在我们必须承认这一过程同时也在不断将有害物质排入穹顶之下,使大气愈加污浊。
如果一个馒头的价格是50分,则单个标准碳氢键价值6.64 x 10-24分。2014年人类消耗的碳氢键总数为1799 x 1037,若换算成货币,则当
年人类活动总值为20万亿美元。按照传统的经济学算法,2014年所谓的全球GDP高达80万亿美元,若按照馒头算法,仅人类活动制造的碳氧键就达40万亿美元。
2014年人类消耗的碳氢化合物和碳氧化合物中碳氢键的数目(1799x 1037)见上图所示(消耗过程产生碳氧键)




No comments yet.